Norine
coelichelin
- Norine ID: NOR01141
- DOI: 10.26097/nor01141
- Family: coelichelin
- Synonym(s):
coelichelin;
- Activity: siderophore
- Category:
peptide
- Formula: C21H39N7O11
- Monoisotopic mass: 565.2707551249999 g/mol
- Source: norine
- Contributor (creation): Norine Team
[CRIStAL (UMR CNRS 9189), ex-LIFL, France, Charles Viollette Institute, ProBioGEM team, Lille, France, University of Lille, France]
- Entry information:
- Type: linear
- Number of monomers: 4
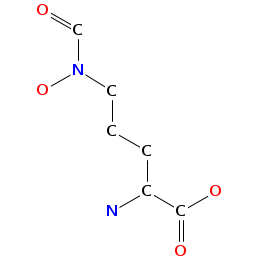
- Smiles: O=CN(CCC[C@H](C(=O)N[C@H](C(=O)O)CCCN(C(=O)[C@@H]([C@H](O)C)NC(=O)[C@@H](CCCN(C=O)O)N)O)N)O
- Graph inference:
- Monomeric composition :
- Graph representation: D-Fo-OH-Orn,D-aThr,OH-Orn,D-Fo-OH-Orn @1 @0,2 @1,3 @2
- Atomic structure:
 vizualisation:
vizualisation:
- Streptomyces coelicolor
- gram: positive
- synonyms: Actinomyces coelicolor,Nocardia coelicolor,Cladothrix coelicolor,Streptothrix coelicolor
- taxid: 1902
(view NCBI taxonomy browser)
- Links between organisms producing the coelichelin: Streptomyces coelicolor, Streptomyces coelicolor
- Exploitation of the Streptomyces coelicolor A3(2) genome sequence for discovery of new natural products and biosynthetic pathways.
Journal of industrial microbiology & biotechnology , 2014, 41(2):219-32
DOI: 10.1007/s10295-013-1383-2
pubMed: 24322202
- MbtH-like protein-mediated cross-talk between non-ribosomal peptide antibiotic and siderophore biosynthetic pathways in Streptomyces coelicolor M145.
Microbiology (Reading, England) , 2007, 153(Pt 5):1405-12
DOI: 10.1099/mic.0.2006/003145-0
pubMed: 17464054
- Discovery of a new peptide natural product by Streptomyces coelicolor genome mining
Sylvie,
Lautru,
Robert,
J,
Deeth,
Lianne,
M,
Bailey,
Gregory,
L,
Challis,
Nature Chemical Biology , 2005, vol. 1,no. 5,pp. 265-269
DOI: 10.1038/nchembio731
pubMed: 16408055
 Return to annotations search
Return to annotations search



 Return to annotations search
Return to annotations search